Closure of patent foramen ovale (PFO) after stroke reduced circulatory total homocysteine (tHcy), a prospective study showed.
Compared with baseline homocysteine values, PFO-related stroke patients with complete closure—about 77% of those who had the procedure—had a significantly decreased tHcy level at median two-year follow-up (10.18 versus 8.98 μmol/l, P=0.007), reported MingMing Ning, MD, MMSc, of Massachusetts General Hospital in Boston, and colleagues in Neurology.
No significant change in post-procedure homocysteine level was observed in patients with either small (10.08 versus 10.38 μmol/l, P=0.60) or moderate-to-large (10.96 versus 13.47 μmol/l, P=0.14) residual shunts.
“In our study, by sampling blood directly from cardiac atrium during PFO closure procedure, we found that PFO-related right-to-left interatrial blood shunting contributes to elevated tHcy level with a ’dose response effect’ such that the degree of shunting correlates directly to the degree of elevation of tHcy levels,” Ning and co-authors wrote.
“Furthermore, tHcy reduction is sustained over the long term after PFO closure in venous blood, whereas medical therapy does not alter tHcy level,” they added.
“tHcy reduction was more dramatic in left atrium than right (P<0.001), suggesting clearance through pulmonary circulation,” they continued. “These findings suggest that PFO itself may participate in clot formation, beyond its role as a conduit for clots.”
Elevated levels of homocysteine have been associated with B vitamin or folate deficiency as well as cardiovascular disease, Alzheimer’s disease, and Parkinson’s disease. Ning’s team agnostically identified biomarkers via mass spectroscopy in a discovery cohort (n=12) and found homocysteine was the most significantly decreased metabolite in intra-cardiac blood following PFO closure. A separate cohort (n=181) was used to confirm this.
“Questions remain about the consequences, if any, of the fairly small tHcy elevation associated with PFO in the data reported here,” noted Padraig O’Suilleabhain, MD, of the University of Texas in Dallas, in an accompanying editorial.
The average tHcy concentration in the cohort with stroke attributed to PFO was 10.45 μmol/l at baseline, which is “within a normal range for most laboratories,” he pointed out. “Complete PFO closure lowered tHcy concentration by approximately 1 μmol/L on average.”
“B vitamin supplementation can lower tHcy about the same amount as PFO closure, so a trial might be designed for secondary prophylaxis of PFO stroke, considering PFO closure is not practical for everyone who might benefit,” he added.
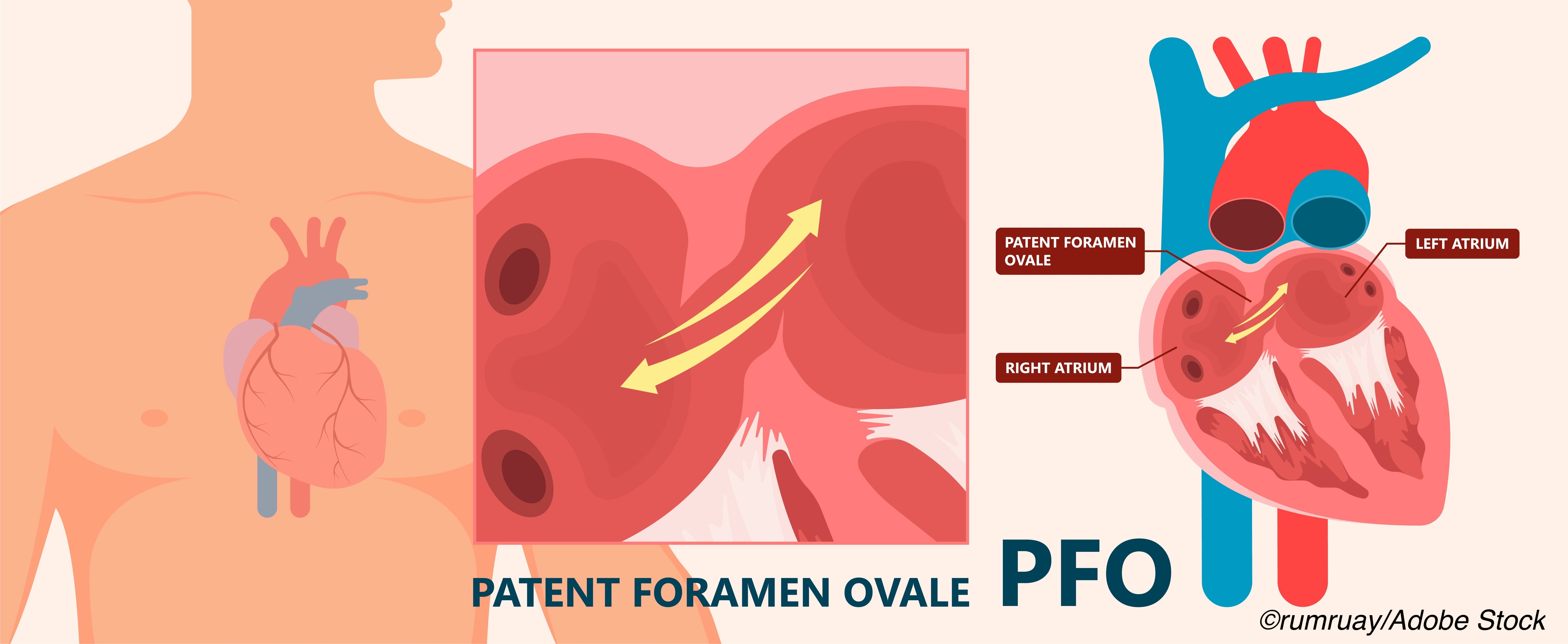
PFO is common—nearly 50% in those with cryptogenic stroke—and has been associated with migraine and stroke. PFO-related strokes are embolic: venous blood entering the right atrium, unfiltered by the lungs passes through the PFO into the left atrium and then into circulation. If the blood bypassing the lungs contains a thrombus, embolic ischemia at the embolus-receiving organ or tissue results. People who have PFO and a tendency to form thrombi have significantly increased stroke risk compared with people who have PFO alone.
A 2020 review described four recent trials consistently favoring efficacy and safety of PFO closure compared with medical therapy alone for stroke prevention, but concluded that “taking into consideration the high prevalence of PFO, the low risk of stroke recurrence under medical treatment and the uncommon yet severe adverse events of the intervention, patient selection is crucial for attaining meaningful clinical benefits.”
PFO-shunted blood may contain factors other than thrombi that are clinically relevant: a 2015 publication suggested an increase in cholesterol efflux from endothelial cells and decrease in lipid oxidation following PFO closure.
“Understanding the blood molecular signature of PFO shunting, critical in heart-brain signaling, may benefit future patient selection for treatment and prevention of PFO-related stroke,” Ning and colleagues observed.
The research team included 254 patients between December 2007 and July 2014 at a single center. All participants were diagnosed with cryptogenic stroke attributable to PFO after other causes of stroke were ruled out.
Residual PFO shunting was predicted by changes in tHcy with respect to baseline, with a receiver operating characteristic curve for discriminating no shunt versus any of small, moderate, or large of 0.659 (95% CI 0.538-0.776, P=0.014). “Biomarkers such as tHcy can potentially serve as cost-effective measures of residual shunting and neurovascular risk for PFO stroke,” the researchers suggested.
“It is unlikely that interventional cardiologists will adopt this in place of a bubble study which has more face validity and less confounding by food intake,” the editorialist observed.
“A greater value of this important report is in opening a new area of research into the role of pulmonary homeostasis into the nexus of PFO and stroke, and potentially migraine and dementia,” O’Suilleabhain continued.
“On a meta level, their fresh take on a pathologic mechanism which has sometimes been taken for granted — paradoxical embolus as presumptive cause of stroke associated with PFO — reminds us to remove the blinkers and look beyond the obvious suspect in our disease models,” he added.
A study limitation is that medications used during PFO closure and in long-term treatment also may have potential influence on tHcy levels, Ning and coauthors acknowledged.
-
Closure of patent foramen ovale after stroke reduced circulatory total homocysteine, a prospective study showed.
-
A change of about 1 μmol/L on average was observed, about the same reduction typically achieved with B vitamin supplementation.
Paul Smyth, MD, Contributing Writer, BreakingMED™
This study was funded by NIH.
Ning reported no disclosures.
O’Suilleabhain reported no disclosures.
Cat ID: 38
Topic ID: 82,38,730,8,38,192,925,159



Create Post
Twitter/X Preview
Logout