A causal classification system applied to randomized trial results discerned subgroups of patients who may benefit from patent foramen ovale (PFO) closure and distinguished them from patients unlikely to receive benefit.
“Among patients aged 18 to 60 with PFO-associated stroke, risk reduction for recurrent stroke with device closure varied across groups classified by their probabilities that the stroke was causally related to the PFO,” wrote David Kent, MD, MS, of Tufts Medical Center in Boston, and co-authors, in JAMA. “Application of this classification system has the potential to guide individualized decision-making.”
For patients classified as unlikely to have PFO involvement in their index stroke (i.e., patients with vascular risk factors and without high-risk PFO features), hazard ratio [HR] was 1.14 (95% CI 0.53-2.46), which was not statistically significant, the researchers reported. The 2-year absolute risk reduction for this group was −0.7%, 95% CI −4.0% to 2.6%.
For patients classified as possibly or probably likely, hazard ratios were statistically significant:
- Possible: HR 0.38, 95% CI 0.22-0.65, with 2-year absolute risk reduction of 2.1% (95% CI 0.6%-3.6%).
- Probable: HR 0.10, 95% CI 0.03-0.35, with 2-year absolute risk reduction of 2.1% (95% CI 0.9%-3.4%).
“The annualized risk of a future stroke for patients assigned to medical therapy was approximately 1%, which accumulates over time; this risk was reduced by device closure by approximately 60%,” Kent and colleagues noted.
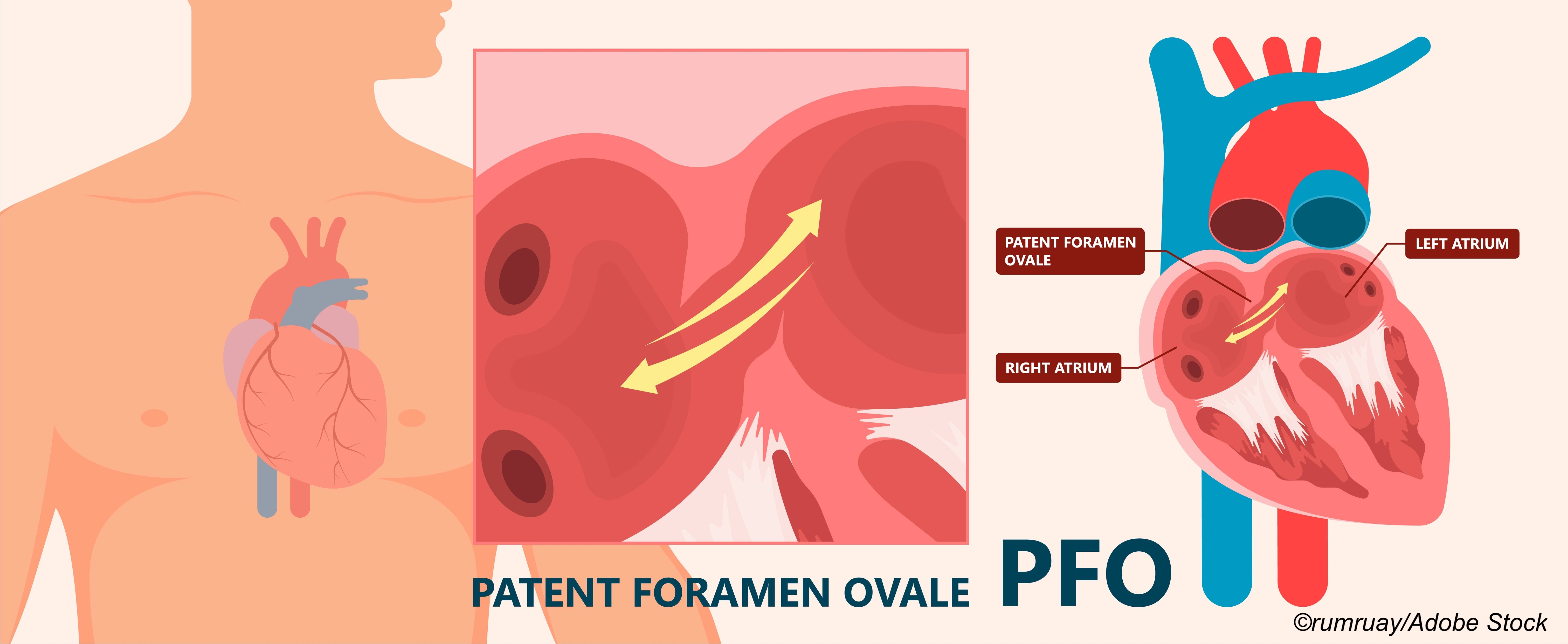
The likelihood of PFO involvement in the index stroke was rated with the PFO-Associated Stroke Causal Likelihood (PASCAL) classification system, which adds high-risk PFO features—atrial septal aneurysm or a large shunt—to a previously studied 10-point Risk of Paradoxical Embolism score (ROPE, with higher scores implying higher risk that PFO was related to stroke).
Based on these data, the group classified the causal role of PFO in the index stroke as unlikely, possible, or probable.
Investigators applied the classification scheme in an analysis of pooled individual patient data (n=3,740; 1,889 had PFO device closure and 1,851 had medical therapy) from six randomized phase III clinical trials conducted between 2000 and 2017.
Median age in both the device closure and medical therapy groups was about 46; males made up 54.2% and 55.9% of the groups, respectively. All participants had an index stroke (or transient ischemic attack) and PFO.
Shunts considered large were reported in 42.9% of the device closure group and 46.8% of the medication only group. Atrial septal aneurysm was considered present in 32.9% and 33.3%, respectively.
Over a median followup of 57 months, there were 121 recurrent strokes. The annualized incidence of stroke with medical therapy was 1.09% (95% CI 0.88%-1.36%) and with device closure 0.47% (95% CI 0.35%-0.65%), with an adjusted HR of 0.41 (95% CI 0.28-0.60).
Absolute risk increases in atrial fibrillation beyond day 45 after randomization in those treated with device closure for the unlikely, possible, and probable PASCAL categories, respectively, were 4.41% (95% CI 1.02% to 7.80%), 1.53% (95% CI 0.33% to 2.72%), and 0.65% (95% CI −0.41% to 1.71%).
“Overall, PFO closure appeared relatively safe,” the authors wrote. “Atrial fibrillation was somewhat more frequent with device closure. However, most atrial fibrillation was transient and did not cause any permanent harm; post-peri-procedural atrial fibrillation was increased by only slightly more than 1% on the absolute scale compared with medical therapy.”
In an accompanying editorial, Robert Yeh, MD, and Doreen DeFaria Yeh, MD, both of Beth Israel Deaconess Medical Center in Boston, noted that the findings “provide critical evidence that can immediately influence the clinical evaluation and care of patients with cryptogenic stroke and PFO.”
Several steps are needed to implement the findings, the editorialists suggested:
- Evaluation of other sources of ischemic stroke by a cardiologist and neurologist. “Exclusion of atrial fibrillation requires prolonged monitoring among patients older than 40 years, an inconvenient but nevertheless essential component before device PFO closure is considered in light of the high rates of undiagnosed atrial fibrillation, estimated at 30% during 36 months of continuous monitoring,” they noted.
- Imaging of the atrial septum, meticulously performed, with advanced imaging (e.g., transesophageal echocardiography, cardiac computed tomography, or magnetic resonance) as needed. “Device closure of PFOs will not reduce stroke risk if a concomitant sinus venosus atrial septal defect is missed and uncorrected,” they wrote.
- Evaluation of factors increasing the likelihood of a causal role for PFO, including proclivity for paradoxical embolization (e.g., hypercoagulability, deep venous thromboses, or pelvic vascular anomalies), presence of atrial septal aneurysm, and large shunt size.
“A multidisciplinary clinical committee can bring together the nuanced expertise of neurologists, hematologists, noninvasive cardiologists, echocardiographers, and structural or congenital heart interventionalists to review all primary data to determine relative risks and benefits of medical therapy vs device closure,” they wrote. “The summative findings of this assessment should be brought back to the patient to facilitate patient-centered shared decision-making.”
“The final decision will remain one associated with some uncertainty,” the editorialists added. “But with new decision tools, as applied by Kent et al, to refine a complex clinical evaluation, physicians will be better equipped to offer more precise individualized recommendations to patients with PFO.”
PFO is common—found in up to 25% of the general population—and PFO-associated strokes account for about 5% of ischemic strokes overall and 10% of ischemic strokes in adults 18 to 60 years old.
“While device closure decreases stroke recurrence risk overall, the best treatment for any individual is often unclear,” Kent and colleagues observed.
The study had several limitations, the researchers noted. The magnitude of the benefit associated with device closure in preventing disabling stroke is unclear, they added. In addition, there was a lack of standard definitions for important variables like ’large shunt’ or the presence/absence of atrial septal aneurysm, which varied across the trials included in the analysis.
-
A causal classification system applied to randomized trial results discerned subgroups of patients who may benefit from patent foramen ovale (PFO) closure and distinguished them from patients unlikely to receive benefit.
-
Application of this classification system has the potential to guide individual patient decision-making, the researchers said.
Paul Smyth, MD, Contributing Writer, BreakingMED™
Research reported in this publication was funded in part through two PCORI awards.
Kent reported receiving grants from the PCORI and WL Gore during the conduct of the study, and speaker honorarium from the Canadian Stroke Consortium outside the submitted work.
Yeh reported receiving grants and personal fees from Abbott Vascular outside the submitted work.
Cat ID: 8
Topic ID: 74,8,730,8,130,192,925



Create Post
Twitter/X Preview
Logout