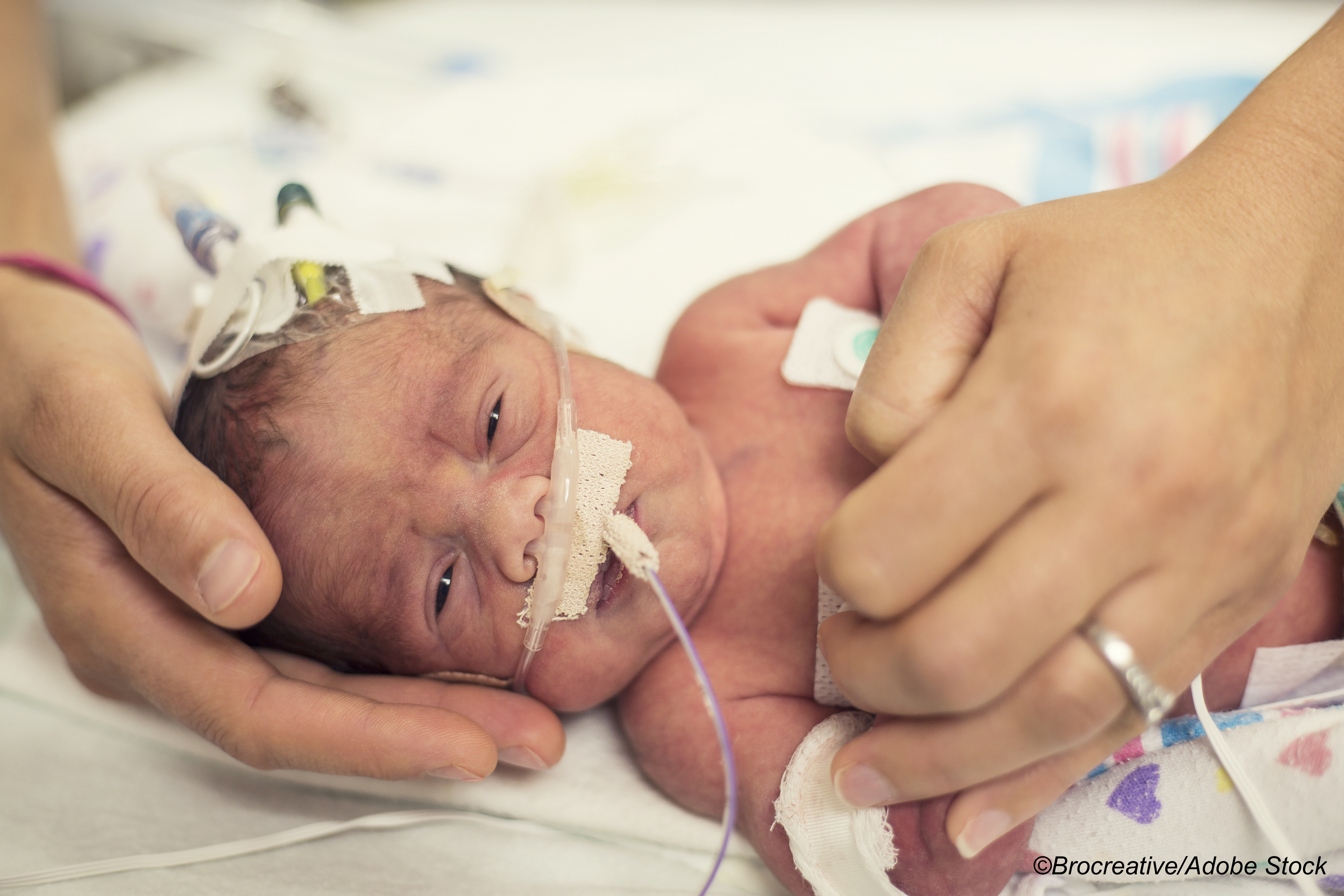
In neonates admitted to newborn intensive care units (NICUs), treating purely electrographic seizures in addition to those that were clinically evident made little difference in death or severe disability outcomes at 2 years, a randomized trial found.
The odds of the primary outcome by age 2—death; a developmental score including motor, language, and cognitive function; or the presence of cerebral palsy, blindness, or deafness—were not significantly different in the electrographic/clinical treatment group compared with the clinical treatment-only group.
The outcome occurred in 44% versus 31%, respectively (OR 1.83, 95% CI 0.96 to 3.49, P=0.14), reported Rod Hunt, PhD, of Monash University in Australia, and co-authors in JAMA Network Open.
“In this randomized control trial, there was little evidence of difference in mortality or morbidity at 2 years of age between an electrographic seizure group and clinical seizure group however, our study was underpowered,” the researchers wrote.
“There was little evidence of a difference in secondary outcomes, including time to suck feeds, seizure burden, or brain injury score,” they added.
Investigators studied NICU patients with at least 35 weeks’ gestation admitted with encephalopathy who were younger than 48 hours old. “Recruitment commenced in March 2012 and ended prematurely at 212 neonates in January 2016 because of slow trial progress and a loss of equipoise at some sites with the publication of Srinivasakumar in October 2015 suggesting that treatment of all electrographic seizures was beneficial,” Hunt and colleagues noted.
Inclusion criteria were neonatal encephalopathy (including coma, stupor, or depressed mental state), hypoxic-ischemic injury (HIE), or suspected neonatal seizures from any cause. All participants had electroencephalographic (EEG) monitoring.
Researchers evaluated 86 newborns in the electrographic/clinical seizure group (in which both electrographic and clinically evident seizure were treated, with the EEG screen visible) and 86 in the clinical seizure group (in which only clinically evident seizures treated, and the EEG screen was covered). A brain injury score was determined by MRI.
The primary outcome was determined at age 2 years as either death; severe disability defined as scores in any developmental domain more than 2 standard deviations below the Australian mean assessed with Bayley Scales of Neonate and Toddler Development, 3rd edition (BSID-III
Electrographic seizures were seen in 84% overall. For the electrographic/clinical and clinical groups, anticonvulsant medications were used in 86% and 69%, respectively.
In total, 9% of neonates in the EEG/clinical seizure group and 4% in the clinical seizure group died before the 2-year assessment (OR 2.66, 95% CI 0.81-8.78). Post-natal epilepsy developed in 6% of the EEG/clinical versus 4% in the clinical group, with no significant difference between groups.
Cognitive scores on the BSID-III were worse in the electrographic/clinical versus clinical-only group, with a significant difference on point scores (mean difference -6.5 points favoring the clinical group (95% CI -1.2 to -11.8, P=0.01).
“Contrary to what we hypothesized, we report an association of improved cognitive outcomes at 2 years in the clinical seizure group,” Hunt and colleagues wrote.
“Most neonatal seizures are subclinical, i.e., EEG epileptogenic discharges occur without any temporally related clinical motor or autonomic symptoms, and, moreover, the diagnosis of clinical seizures in neonates is highly unreliable,” wrote Martin Offringa, MD, PhD, and Brian Kalish, MD, both of the University of Toronto in Canada, in an accompanying editorial.
“Therefore, neuromonitoring with amplitude-integrated EEG (aEEG) or continuous EEG (cEEG) has become the standard of care among tertiary NICUs,” Offringa and Kalish continued. “Yet, while EEG monitoring in the NICU enables precision-guided approaches to diagnosis and therapy, an unresolved question in neonatology and neonatal neurology is whether the treatment of subclinical electrographic seizures is beneficial.”
“This is the largest study to date comparing two current clinical approaches to neonatal seizure management in the NICU,” the editorialists pointed out. “The inclusion of patients at an early stage in the disease with a range of seizure etiologies is highly pragmatic and reflects clinical practice, in which the precise cause of the seizures is often unknown at the time of treatment initiation.”
Given uncertainties about treatments, including the efficacy of phenobarbital (often considered first line treatment for neonatal seizures), “it is imperative that we rationalize the dose and duration of anticonvulsant administration during critical periods of neurodevelopment, and that our management decisions are based on well-executed studies that show the drug interventions do more good than harm,” they added.
Seizures in newborns are associated with increased risk of death and neurodevelopmental disability and are often treated aggressively. The causes differ for term infants (HIE, stroke, malformations, metabolic changes) versus pre-term infants (intraventricular hemorrhage and infections), and their clinical manifestations differ considerably from later-life semiology. Uncertainty remains as to best treatments and tradeoffs between adverse effects of seizure versus their treatment.
Limitations of the trial include the fact that recruitment was stopped early and that the study was underpowered. Also, the study used a compressed 1-2 channel EEG representation (aEEG) different from full-channel cEEG, which limits generalizability for settings using the more resource-intensive cEEG monitoring.
-
Treating purely electrographic seizures in addition to those that were clinically evident in neonates made little difference in death or severe disability outcomes at 2 years, a randomized trial found.
-
The odds of the primary outcome by age 2—death; a developmental score including motor, language, and cognitive function; or the presence of cerebral palsy, blindness, or deafness—were not significantly different in the electrographic/clinical treatment group compared with the clinical treatment-only group.
Paul Smyth, MD, Contributing Writer, BreakingMED™
This study was funded by the National Health and Medical Research Council of Australia.
Hunt had no disclosures.
Offringa and Kalish had no disclosures.
Cat ID: 34
Topic ID: 82,34,730,34,138,192,925



Create Post
Twitter/X Preview
Logout